|
(Glucose transporters transport only free glucose, not phosphorylated glucose) Reaction 2: Isomerization of glucose-6-phosphate to fructose 6-phosphate. 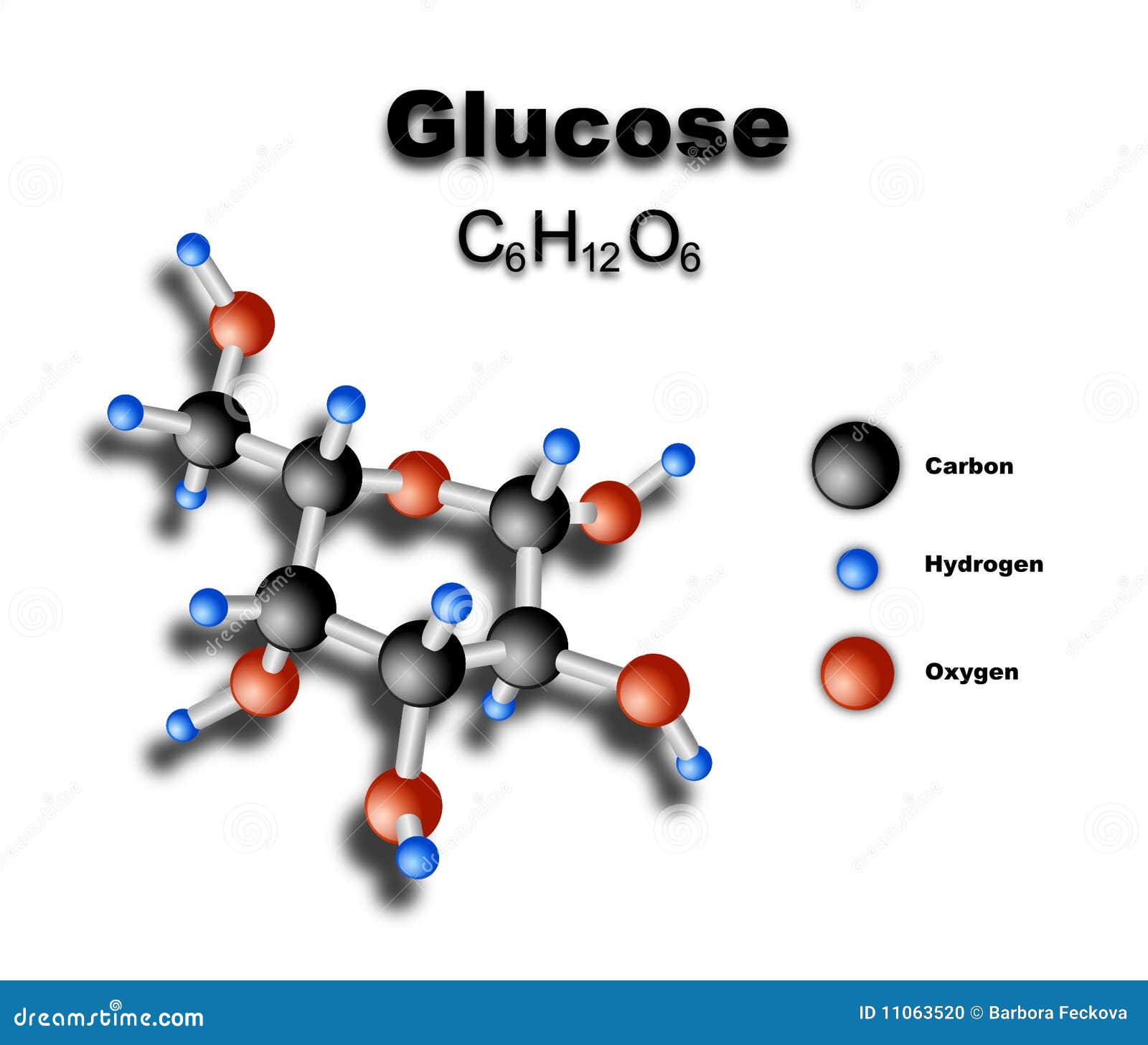
When they give them up to become stable, they will have a negative charge and the recipient will have a positive charge since one has more electrons than protons and vice versa. b- May be non carbohydrate e.g glycolipid, glycoprotein. When they just release it to another element, that is an ionic bond. Sucrose is composed of glucose and fructose which is shown in Fig. 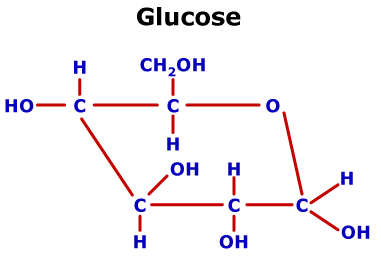
(monosaccharide monosaccharide)hemiacetal hemia cetalacetal. So the glucose gets trapped inside the cell. OH of other-2 compound : A- May be carbohydrate ( Monosaccharides can be linked to each other by O- glycosidic bonds to form disaccharides, oligosaccharides and polysaccharides). Ask your healthcare team for recommendations on structuring your blood glucose monitoring plan. Fortunately for Fischer, many different pentoses and hexoses already were available from the efforts of earlier investigators, and the principles of optical isomerism were well understood as the result of the work of van't Hoff. It has a low Km for glucose thus, once glucose enters the cell, it gets phosphorylated. What you will be unable to fully appreciate is the great difficulties of working with carbohydrates - that is, their considerable solubility in water, instability to strong oxidizing agents and acidic or basic reagents, reluctance to crystallize, and their tendency to decompose rather than give sharp melting points. The overall mixture is laevorotatory and this is because the laevorotation of fructose (-92.4) is more than the dextrorotation of glucose ( 52.5). The complete logic of Fischer's procedures for determination of the configuration of glucose is too involved to be explained here in detail. Sucrose being dextrorotatory in nature gives dextrorotatory glucose as well as laevorotatory fructose on hydrolysis. Therefore natural glucose is specifically \(D\)-glucose: Since cellulose is a homopolymer of a glucose derivative, it is a great source of fermentable. \) is the same as the configuration of the simplest "sugar" \(D\)-\(\left( \right)\)-glyceraldehyde, \(3\), (see Section 19-5).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
